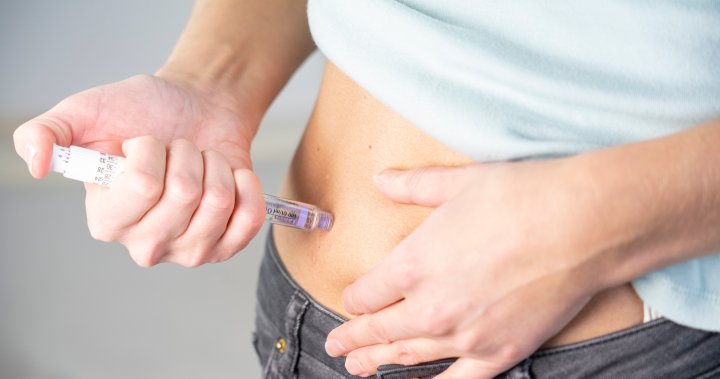
As drugs like Ozempic and Wegovy rise in popularity, there have been increased calls about injectable drugs often used for weight loss to some poison control centres in parts of Canada, according to data gathered by Global News.
These come from people experiencing adverse side effects from the medications, or even what some officials consider an “overdose.” Global News reached out to Canada’s provinces and territories about whether there was a rise in calls to poison control centres amid increased calls reported in the U.S.
At least three provinces — Ontario, Quebec and Manitoba — have each reported an increased number of calls involving semaglutide, Ozempic’s generic name, to its poison control centres this year, with each seeing more than a 50-per-cent increase compared to 2022, though the numbers do not specify if the calls are from diabetes users or those taking it for weight loss.
In Ontario and Manitoba, the tallies include reports for both semaglutide and liraglutide, which is used to treat diabetes but has also been approved for weight-loss use, and neither provided further breakdowns.
Manitoba, according to Shared Health, saw a 200-per-cent increase between 2022 and 2023, though the numbers themselves remain low with calls rising from two to six. Those numbers had also been at two and zero per year dating back to 2019.
Quebec has seen a gradual increase since 2020, when 13 calls were reported, with a more than 80-per-cent jump this year, with 54 calls.
The province also noted that the majority of cases each year were therapeutic errors — often considered an error in dosing or in dispensing, according to the Journal of Pharmaceutical Research International. In 2023, 33 cases were classified as such while the province noted the remainder were mainly “intentional overdose and intentional misuse.”
And in Ontario, the number of calls jumped by about 55 per cent to 84 in 2023 from 54 in 2022, after a gradual increase the previous three years, though the Ontario Poison Centre advised the calls were related to not just semaglutide but also liraglutide, another diabetes medication.
It also notes that the numbers may underrepresent the total number of potential overdoses, as calls to poison control centres are voluntary.
The Atlantic Canada Poison Centre said it has seen fewer than five calls in the past 24 months and has not seen an increase year-over-year.
An official with Alberta Health told Global News on background that its Poison and Drug Information Service had 23 calls related to semaglutide/Ozempicin the past six months and that the province has seen a decrease compared to previous years. They did not provide yearly breakdowns.
Saskatchewan, meanwhile, deferred to the Poison and Drug Information Service (PADIS) which recommended calling a toll-free number that was out of service.
Global News did not hear back from the province of B.C. or the three territories by publication.
Novo Nordisk, the maker of diabetes drugs Ozempic and Rybelsus and its other drug specifically for obesity, Wegovy, told Global News in an emailed statement Monday that patient safety is of “utmost importance.”
The company said it is monitoring the safety profile of their products and is collaborating with health authorities to ensure patient safety information and adequate details of side effects are included in the product monograph. It added anyone prescribed any of the three drugs should have a detailed discussion with their physician to ensure proper medication use.
Jen Belcher, vice-president of strategic initiatives at the Ontario Pharmacists Association and a pharmacist herself, told Global News in an interview that a rising rate of calls about dosing errors is not unexpected given the total number of people using Ozempic has increased “quite dramatically” in the past several years.
“With that overall increase in the total number of prescriptions being dispensed, the number of people who might have errors related to dose or be seeking assistance, you know, that absolute value, of course will increase as well,” she said.
Get the latest Health IQ news.
Sent to your email, every week.
The drug has been billed as revolutionary for the treatment of Type 2 diabetes, but its demand has been surging amid popularity among celebrities as a weight-loss solution.
As a result, it has prompted shortages of the drug both in the U.S. and Australia, and at the start of this year, some consumers south of the border have even been turning their eyes to Canada as a cheap place to get the out-of-stock medication. Now, 12 months later, there’s shortages in both countries.
The Ozempic “pen” is typically used as a weekly medication and is administered by the patient who selects their dose by turning the selector until the dose counter shows the amount needed.
The pens range from a red-label pen with 0.25 and 0.5 milligrams (mg), a blue-label pen with one mg, or a two-mg pen with a yellow label. A patient then injects the needle, which is attached to the pen by the user, into the skin and held down until the dose counter reaches zero.
While the pen contains a dose counter and there are a typically a set number of clicks for each dose, Belcher said getting the wrong dose can happen.
“There’s always the possibility that you go beyond or you’re perhaps not paying attention as you do it or if something else is occurring where you get confused, you think you’re moving up to 0.5mg but in reality, it was supposed to be another period of time for which you are on the lower dose,” she said.
Belcher also noted that in some cases, a patient may get to the end of the week but didn’t track when they had their dose initially and then, “out of an abundance of caution,” takes another dose.
Dr. Sean Wharton, an internal medicine physician, weight management specialist and medical director of the Wharton Medical Clinic, told Global News that while there has been a number of calls to these centres, he believes it is “not concerning.”
He said because the drugs, which are relatively new and people might not know how to use them properly, are seeing greater uptake, there is an “expected increase” in side effects.
The benefit, however, is it can create more discussion about the drugs.
“It’s not an overdose, it’s a side-effect profile,” he said.
He went on to note that from his perspective, an overdose would be something severe enough “that it’s going to cause a harm to life or a significant issue,” but suggested that’s not what is happening here.
Wharton noted that many of the calls he’s received from his own patients report side effects “you could call your family doctor about,” such as increased gastrointestinal symptoms.
“If you ramp up in the dose too quickly, then you’ll get more of those side effects. If you’re more sensitive to it, you’ll get more of those side effects,” he said.
According to Ozempic’s own website, the most common side effects associated with the drug can be nausea, stomach pain, constipation, diarrhea and vomiting.
Dr. Tom Elliott, medical director of BCDiabetes and LifestyleRx, told Global News in an interview while he wouldn’t call the side effects an “overdose” or even “poisoning,” the symptoms some people may be facing are a result of how Ozempic is dose-dependent.
“So if you give yourself four times the dose, which is conceivable … that could result in unexpected nausea,” he said.
He noted that what patients may face if they take more than the recommended dose may be a case of severe side effects that, while not life-threatening, can be “nasty.”
However, the increase of calls to poison-control centres, he believes, could also be due to some patients not being connected to their pharmacist or primary-care provider.
“That’s what I think is happening, it’s people who just haven’t been told about nausea and vomiting. It’s severe and they’re calling and they need help,” he suggested.
When it comes to an overdose or incorrect use of the drug, another issue that is faced by patients is the half-life of the drug sitting at about seven days, meaning it takes one week to clear half of it from your body.
Wharton notes the half-life of the drug means while it may allow you to give people a bit of extra time to take the weekly dose if they miss the usual date of injection — you may be able to take it within four days after that usual date — it also means if you’re having side effects due to the actual dose, it may take a longer time for them to disappear.
“So the big challenge for many people is, why would you take yet another dose and do so knowing that you have significant side effects previously,” he said. “You’ll go up on the dose and take it yet again, so the side-effect will be significant.”
Elliott suggests if a person is experiencing significant symptoms, medical advice should still be sought, but when it comes to taking your next dose of the drug it may be best to wait until your symptoms go away and then go down to the “last tolerated dose.”
However, he notes with Ozempic and similar medications, side effects still happen, and thus medical professionals need to ensure patients are aware.
“It’s incumbent on the prescribing physician to mention them and to give them sufficient weight so the patient knows that, you know, is this something that’s critical, is it something that’s not so critical,” he said.
Canada is not the only country facing an increase in calls involving products containing semaglutide, including Ozempic and Wegovy.
In the U.S., poison control centres have documented 2,941 exposures to products from January to November, an increase nearly 15-fold from 2019 when just 196 calls were received.
A statement to Global News from America’s Poison Centers Monday said the exposures included two cases documented as involving a compounded injectable product containing semaglutide, both reported in November, which can lead to adverse effects.
Compounded drugs are created through the process of combining, mixing, or altering ingredients to create a medication tailored to the needs of an individual patient, according to the U.S. Food and Drug Administration (FDA).
Such compounding can occur when a drug is in shortage, and compounders — which can sometimes be pharmacies — may be able to provide such a version if they meet certain requirements in the federal Food, Drug and Cosmetic Act.
The FDA notes it has received adverse event reports after patients used compounded semaglutide, including ones that may be using salt forms of the generic drug, adding the drugs have not been shown to be safe and effective.
America’s Poison Centers told Global News that due to data limitations on the national level, it is unable to distinguish all cases of compounded semaglutide from other sources and as cases are voluntarily reported, the data may underestimate the number involving this form of the drug.
Compounded semaglutide is less likely to be seen, at least from qualified pharmacies themselves in Canada, Belcher notes, as laws prohibit pharmacies from compounding or making medication that is commercially available.
While there may be concerns over the rise in calls to poison control centres in both countries, Wharton said with more use will come improvements to dosing.
“So just like anything that becomes popular really quickly, the internet, artificial intelligence, we’re going to see some challenges,” he said.
“Eventually, we hope that things end up getting better because we have the capacity as human beings to understand how to make things better and how to mitigate it.”